Aurobindo Pharma Ltd - Multiple Openings for Regulatory Affairs -
Formulations
Tuesday 9 February 2021
Aurobindo Pharma Ltd - Multiple Openings for Regulatory Affairs - Formulations
Monday 8 February 2021
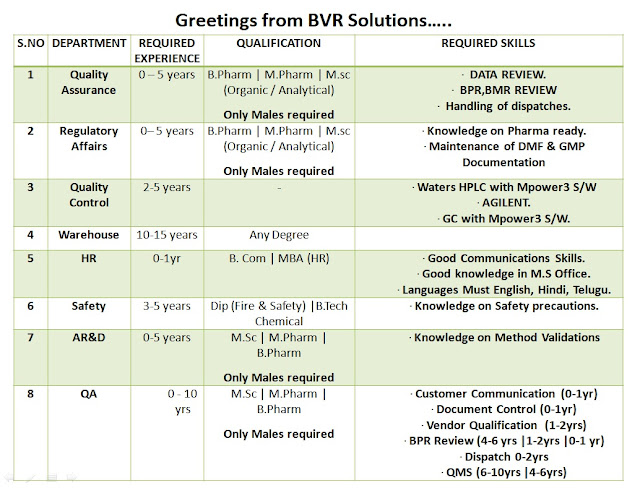
Walk-In Interviews for Freshers & Experienced in QA / QC / Regulatory Affairs / AR&D / Production / HR Departments - API Bulk Drug Plant on 9th to 13th Feb' 2021
Dear Aspirants,
Greetings from BVR Solutions…..
We are looking for Fresher's & experienced candidates with good
communication skills for API BULK DRUG PLANT based @ Hyderabad.
Candidates are requested to carry recent passport size photo, updated
resume, photo copies of Educational Certificates.
Contact: 9121514444|8185999958.
040-35115211
Interview date: 09-02-2021 to 13-02-2021
Timings: 9.30 am
Address: BVR SOLUTIONS, plot no: 60 1st floor 103, near
arjun theatre (opposite to siri tiffins), jalavayu vihar road, western hills
KPHB, Hyderabad
Friday 5 February 2021
Naprod Life Sciences Pvt. Ltd - Openings for Regulatory Affairs / SCM / Business Developments / Accounts - Apply Now
Naprod Life Sciences Pvt. Ltd - Openings for Regulatory Affairs / SCM /
Business Developments / Accounts - Apply Now
Thursday 4 February 2021
Daily Walk-In Interviews for Freshers & Experienced in QA / QC / Regulatory Affairs / Warehouse / HR / Safety / AR&D Departments - Bulk Drug Plant - Apply Now
Dear Aspirants,
Greetings from BVR Solutions…..
We are looking for Fresher & Experienced candidates with good
communication skills for API BULK DRUG PLANT based @ Hyderabad.
Candidates are requested to carry recent passport size photo, updated
resume, photo copies of Educational Certificates, 10th original
certificate.
Contact:
9121514444 | 8185999958.
040-35115211
E-mail
id: bvrcareers@gmail.com. / hr.bvr09@gmail.com
Address: BVR SOLUTIONS, Plot no: 60, 1st floor 103, Near
Arjun Theatre (opposite to Siri Tiffins), Jalavayu Vihar road, western hills,
KPHB, Hyderabad...
SUN PHARMA LTD - Walk-In Interviews for Regulatory Affairs on 7th Feb' 2021
Walk in Interview: Regulatory
Affairs (Formulation ROW/EM) Sun Pharma - Baroda
Date: Sunday, 7th February | Time: 10.00 am - 12.00
noon.
Role: Sr. Officer / Executive / Sr. Executive
Qualification: B.Pharm / M.Pharm with excellent
academic track record.
Experience: 2 - 6 years
Venue of Interview: Sun Pharma, Near Akshar Chowk,
Tandalja Road, Baroda
1) Job
Description: - RA EM
- Regulatory submission (i.e. new and post
approval variation) in key emerging markets like Brazil & LATAM,
ASEAN, R/CIS & MENA region.
- Better understanding on regulatory guidance
which helps to prepare good quality dossiers thereby enabling faster
approvals & timely launch;
- Also ensures business continuity through timely
life-cycle Management.
2) Job
Description: - RA - US & OAM (Formulation)
Dossier submission for US /OAM group preferably having experience in
Injectable dosage forms.
Who should not attend: Candidates with experience in API,
India Market and Freshers
Candidates who are unable to attend may send in their resumes to: Hr.tandalja@sunpharma.com
IMPORTANT NOTE: Sun Pharma does not seek payment of any kind from a
prospective candidates for employment with Sun Pharma or authorize any agency
or any individual to collect or charge any fees or charges for recruitment.
Please be cautious while dealing with any recruitment agency or anyone asking
you to pay money representing himself to be representing Sun Pharma.
Sunday 31 January 2021
70 Openings | Walk-In Drive for Regulatory Affairs on 6th Feb' 2021 @ MSN Laboratories Pvt. Ltd
Walk- In
Drive for Regulatory Affairs Dept. in API Division- R&D Center.
Job Description: (Regulatory Affairs - API Division)
Should have minimum of 1 to 8 years of experience in API Regulatory Affairs
Job Title: Executive / Senior Executive Experience: 1 to 8 years in API RA
field
Education: B Pharma / M Pharma / M.Sc
Department: Regulatory Affairs
Job Title: Executive / Senior Executive Experience: 1 to 8 years in API RA
field Education: B Pharma / M Pharma / M.Sc
Department: Regulatory Affairs
Job Profile:
1. Collecting and reviewing the documents received from various departments
R&D, AR&D, QA, QC and Production etc at each and every stage of
manufacturing of drug substances to minimize the errors at the time of submission
to regulatory agencies.
2. Hands on experience on the preparation of DMFs and their registration
processes in the major regulatory regions [US, Canada, Europe etc]..
3. Hands on experience on preparation of drug master files, Applicants Parts,
Tech Pack, life cycle management [updates and amendments] and drafting of
response to deficiencies and customer DMF review comments for all global
regions.
4. Should have hands on experience on eCTD regulatory submission tools.
5. Should have knowledge on the requirements to respond to the deficiencies.
6. Participation in the cross functional team meetings and providing regulatory
inputs to the Product Development Teams.
Job Title: Junior Manager / Assistant Manager Experience: 1 to 8 years in API
RA field Education: B Pharma / M Pharma / M.Sc
Department: Regulatory Affairs
Job Profile:
1. Good scientific conceptual background to the level to evaluate the processes
and procedures in the area of Quality, R&D, AR&D and Production.
2. Manageable communication in English [should be able to manage regulatory and
scientific discussions in the internal and external meetings and one to one
interactions within the organization and with customers]
3. Manageable knowledge on the regulatory and quality guidelines from various
regulatory agencies ICH, FDA, EMA, Health Canada, ANVISA, PMDA, MFDS, CFDA etc.
4. Thorough knowledge and hands on experience on the DMF registration and
marketing authorization application systems in the major regulatory regions
[US, Canada, Europe etc]..
5. Hands on experience on preparation of drug master files, life cycle
management and drafting of response to deficiencies and customer DMF review comments
for all global regions.
6. Should have knowledge on product developmental and the quality systems to
ensure the regulatory compliance.
7. Should have hands on experience on eCTD regulatory submission tools.
8. Conducting meetings for the deficiencies received from various regulatory
agencies and should have knowledge on the requirements to respond to the
deficiencies. Ensuring that the accurate and adequate responses are sent to the
authorities within the timelines defined by agencies.
9. Thorough review and ensuring that the regulatory submissions are adequate
and error free with minimum open issues and closing of open issues before
filing or before the receipt of deficiencies.
10. Participation in the cross functional team meetings and providing
regulatory inputs to the Product Development Teams.
11. Reviewing the documents received from various departments R&D,
AR&D, QA, QC and Production etc at each and every stage of manufacturing of
drug substances to minimize the errors at the time of submission to regulatory
agencies.
12. Review and assessment of change controls and providing guidance to the team
on proposed changes.
13. Ensuring that the regulatory databases are properly maintained and updated
on time to time for each regulatory activity.
Drug Regulatory Affairs API RA Dept.
Exp: 1 to 8
Years
Position: Junior Executive / Executive /Senior Executive / Junior Manager
Date of Interview:
06.02.2020 ( Saturday )
Interview Time:
9.00 AM to 1.00 PM
Venue Details:
MSN Laboratories Pvt. Ltd.,
R&D Center, Pashamylaram
Ph No : +91-8452304799
040-30438786
Work Location : MSN LS II & R&D Center
Wednesday 27 January 2021
Lincoln Pharmaceuticals Ltd - Urgent Openings for Regulatory Affairs - Apply Now
Lincoln Pharmaceuticals Ltd - Urgent Openings for Regulatory Affairs -
Apply Now
Walk-In Interviews for Freshers & Experienced in QA / QC / Regulatory Affairs / AR&D / Maintenance / Safety / Administration - API Bulk Drugs Plant on 27th Jan' to 7th Feb' 2021
Dear Aspirants,
Greetings from BVR Solutions…..
We are looking for Fresher
& Experienced candidates with good communication skills for API BULK DRUG
PLANT based @ Hyderabad & Vizag locations.
Candidates are requested to
carry recent passport size photo, updated resume, photo copies of Educational
Certificates, 10th original certificate.
Contact: 9121514444 | 8185999958 | 7337227575 | 8367563333
E-mail id: bvrcareers@gmail.com
/ hr.bvr09@gmail.com
Address: BVR SOLUTIONS,
Plot no: 60 1st floor,
103, Near arjun theater (opposite to siri tiffins),
Jalavayu vihar road, western
hills, KPHB, Hyderabad